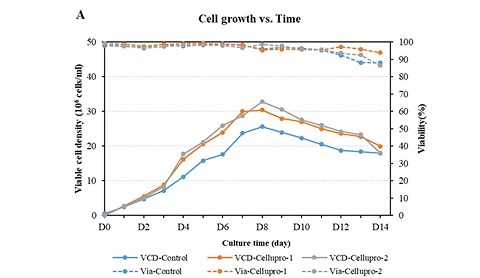
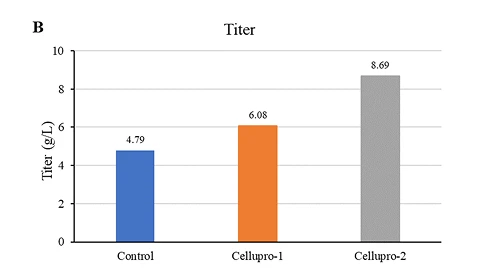
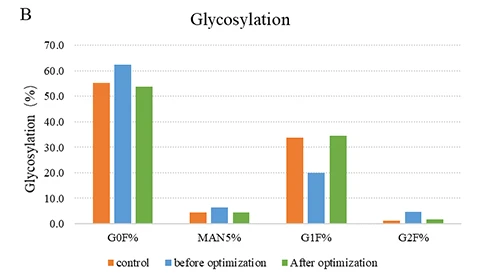
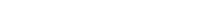Figure (A): While maintaining consistency with the original process, CelluPro customized media significantly increased the viable cell density (VCD) and peak VCD of the client‘s original process, and maintained good cell viability (Via) in the late culture stages.
- About CelluPro +
- Products +
- Services +
- News Center +
- Contact Us +