- About CelluPro +
- Products +
- Services +
- News Center +
- Contact Us +
Mar.09, 2021
On March 9, 2021, the National Medical Products Administration of China approved Telitacicept, a Class 1 innovative drug developed by RemeGen, for the treatment of systemic lupus erythematosus. This marks the first domestically developed new drug for systemic lupus erythematosus approved in China in over 60 years, bringing new hope to patients with lupus in the country.
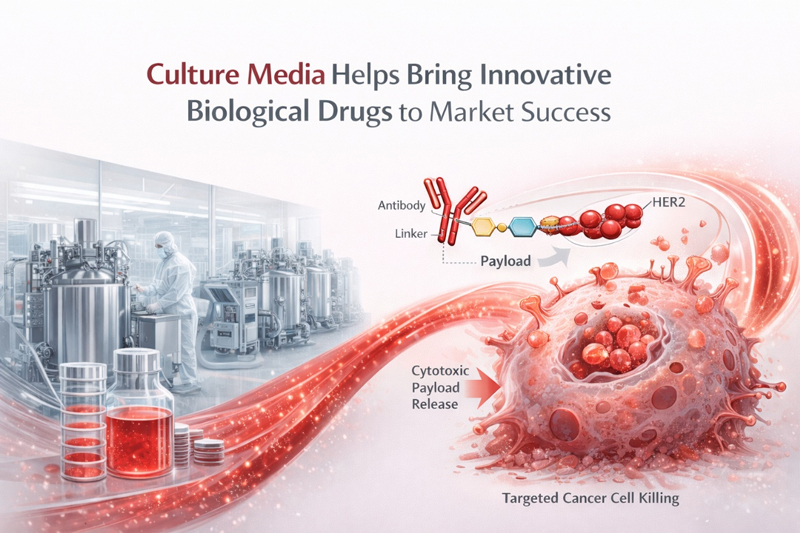
Telitacicept is a TACI-Fc fusion protein that targets two key cytokines, B-lymphocyte stimulator (BLyS) and a proliferation-inducing ligand (APRIL), which regulate the division and differentiation of B lymphocytes. With its novel structure and dual-target mechanism, Telitacicept effectively inhibits the maturation and differentiation of B cells, reducing immune responses and achieving therapeutic effects for autoimmune diseases.
The approval of Telitacicept signifies that CelluPro, Ltd. has become one of the first domestic culture media brands to contribute to the successful market approval of a domestically developed innovative biological drug. CelluPro, as a long-term culture media supplier to RemeGen, extends its sincere congratulations on this achievement.
