- About CelluPro +
- Products +
- Services +
- News Center +
- Contact Us +
Jun.09, 2021
On June 9, 2021, China's National Medical Products Administration (NMPA) announced the approval of Disitamab Vedotin (Brand Name: Aidixi®, Research Code: RC48), the country’s first original antibody-drug conjugate (ADC) new drug. It is approved for the treatment of patients with HER2-overexpressing locally advanced or metastatic gastric cancer (including gastroesophageal junction adenocarcinoma) who have received at least two prior systemic chemotherapy regimens. The approval of Disitamab Vedotin breaks the previous absence of original domestic ADC drugs in the field and fills a global treatment gap for HER2-overexpressing gastric cancer patients in later-line settings, marking a significant milestone in the history of China's innovative biopharmaceutical development.

Aidixi is an innovative antibody–drug conjugate (ADC) independently developed by RemeGen. ADCs are composed of a monoclonal antibody linked to a small-molecule cytotoxic agent, enabling precise targeting and destruction of cancer cells. Unlike similar ADCs developed overseas, Disitamab Vedotin incorporates a novel antibody with differentiated biological characteristics. It specifically targets the HER2 protein expressed on the surface of tumor cells, enabling precise recognition, efficient internalization, and subsequent release of the cytotoxic payload to eliminate cancer cells. As a result, it demonstrates enhanced targeting capability, improved therapeutic efficacy, and a more favorable resistance profile.
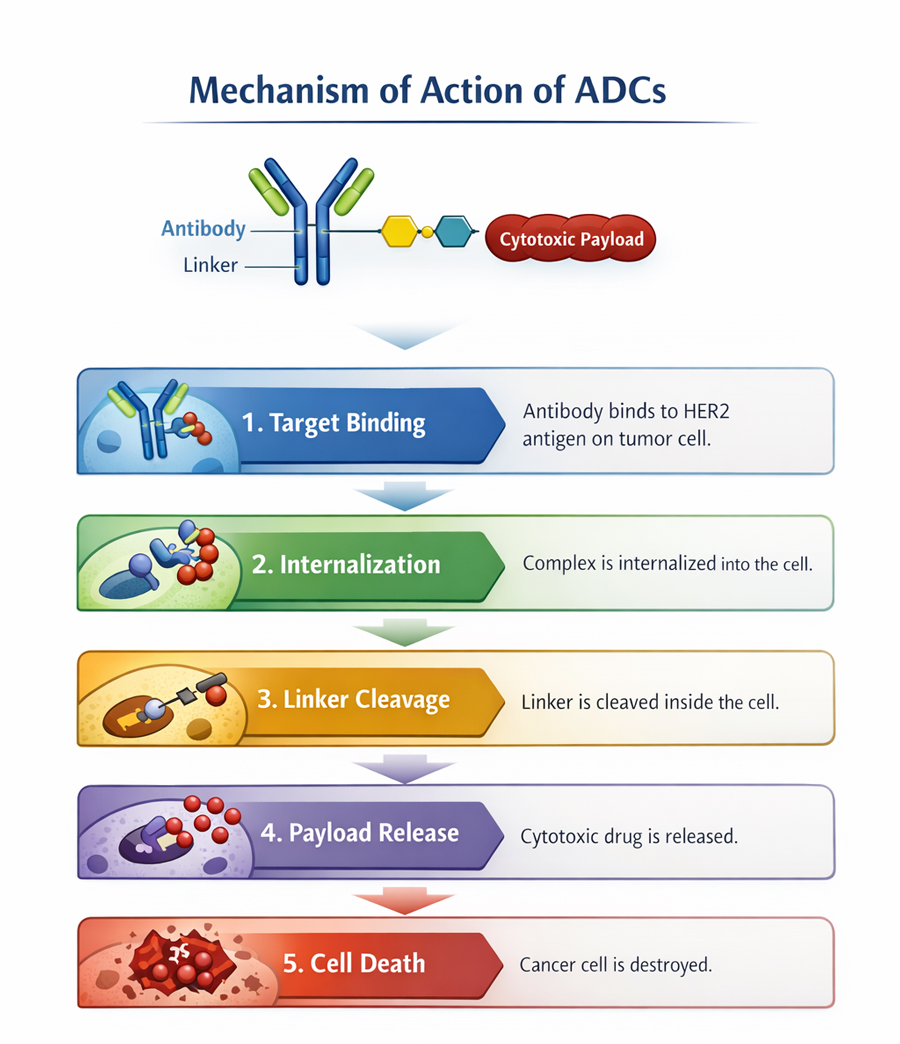
Clinical data indicates that Disitamab Vedotin is globally advanced in ADC clinical trials for HER2-expressing gastric and urothelial cancers. Multiple studies are also ongoing in breast cancer, addressing several gaps both in China and internationally. In August 2020, its New Drug Application for the gastric cancer indication was granted priority review by the NMPA. Internationally, Disitamab Vedotin has received Breakthrough Therapy Designations from both China's NMPA and the U.S. FDA for the urothelial cancer indication.
With the approval of Aidixi, CelluPro, Ltd. (CelluPro) becomes the first Chinese culture media brand to have enabled the successful launch of a domestically developed ADC new drug. As a long-term culture media supplier to RemeGen, CelluPro extends its sincere congratulations on this significant achievement.
